Atmospheric Pressure
Atmospheric pressure is the pressure exerted by the air on all surfaces within the atmosphere. It results from the weight of the column of air above a given point. It is denoted by \( p_{at} \). In the International System of Units (SI), the standard value is \[ p_{at} = 1.013 \cdot 10^5 \ \text{Pa} \] This quantity is known as standard (or normal) atmospheric pressure and, by convention, corresponds to the mean pressure measured at sea level, equal to 101 325 Pa.
The SI unit of pressure is the pascal (Pa).
$$ 1 \ Pa = 1 \ \text{N}/\text{m}^2 $$
Since the atmospheric pressure (pat) is
\[ p_{at} = 1.013 \cdot 10^5 \ \text{Pa} \]
and \( 1 \ \text{Pa} = 1 \ \text{N}/\text{m}^2 \), we may write, to a good approximation,
\[ p_{at} \simeq 10^5 \ \text{Pa} = 10^5 \ \text{N}/\text{m}^2 = 10 \ \text{N}/\text{cm}^2 \]
This approximation has an immediate physical interpretation. On each square centimeter, the atmosphere exerts a force of about 10 N, approximately equal to the weight of a 1 kg mass under Earth’s gravity.
Equivalently, atmospheric pressure corresponds to a force per unit area comparable to supporting roughly 1 kg on every cm² of surface.
Other Units of Measurement
In meteorology, pressure is commonly expressed in hectopascals (hPa).
$$ 1 \ hPa = 10^2 \ Pa $$
Historically, the bar was also widely used.
$$ 1 \ bar = 10^5 \ Pa $$
Therefore, standard atmospheric pressure is approximately equal to 1 bar:
$$ p_{at} \approx 1 \ bar $$
Measurement of atmospheric pressure. Instruments used to measure atmospheric pressure are called barometers, a term that reflects the older unit, the bar. Barometers determine air pressure either through mechanical effects, as in the mercury barometer, or through elastic deformation, as in aneroid barometers.
Dependence on Altitude and Weather
Atmospheric pressure is not constant. It decreases with increasing altitude because the overlying air column becomes thinner, and it varies continuously with meteorological conditions.
The value \( 1.013 \cdot 10^5 \ \text{Pa} \) should therefore be regarded as a reference value rather than a universal constant.
Why Don’t We Feel Atmospheric Pressure?
Although atmospheric pressure is large, it is not directly perceived by the human body.
This follows from a fundamental property of fluids at equilibrium: at any given point, pressure acts equally in all directions.
As a consequence, air pressure exerts forces of equal magnitude and opposite direction on the surfaces of our body.
For example, if a hand is held horizontally, the air exerts equal forces upward and downward. If the hand is oriented vertically, equal and opposite horizontal forces act on its sides. In every configuration, the resultant force is zero. The forces due to atmospheric pressure cancel out, which explains why we do not “feel” the pressure.

A Practical Example
Consider the force exerted by atmospheric pressure on the palm of a hand.
Assume, for simplicity, that the palm is rectangular with dimensions \( 10 \ \text{cm} \times 7 \ \text{cm} \).
Given:
$$ p_{at} = 1.0 \cdot 10^5 \ \text{Pa} $$
and the palm area:
$$ A = 10 \cdot 7 = 70 \ \text{cm}^2 = 7.0 \cdot 10^{-3} \ \text{m}^2 $$
The force exerted by a pressure on a surface is:
$$ F = p \cdot A $$
Substituting:
$$ F = (1.0 \cdot 10^5) \cdot (7.0 \cdot 10^{-3}) = 7.0 \cdot 10^2 \ \text{N} $$
$$ F \approx 700 \ \text{N} $$
The equivalent mass associated with this force is obtained from \( F = m g \), with \( g \approx 9.81 \ \text{m/s}^2 \):
$$ m = \frac{F}{g} $$
$$ m = \frac{700 \ \text{N}}{9.81 \ \text{m/s}^2} \approx 71 \ \text{kg} $$
Thus, the force is comparable to the weight of a 71 kg mass.
Nevertheless, the hand is not crushed because the same pressure acts on the opposite side with equal magnitude and opposite direction, resulting in mechanical equilibrium.

This example underscores a key principle: observable mechanical effects arise from pressure differences, not from the absolute value of pressure.
Examples
Example 1
Calculate the force exerted by atmospheric pressure on the water surface in a glass with a diameter of 10 cm.
Pressure formula:
$$ p = \frac{F}{A} $$
Force due to pressure:
$$ F = p \cdot A $$
Atmospheric pressure:
$$ p \approx 1.01 \times 10^5 \ \text{Pa} $$
Glass diameter:
$$ d = 0.10 \ \text{m} $$
Radius:
$$ r = 0.05 \ \text{m} $$
Surface area:
$$ A = \pi r^2 = \pi \cdot (0.05)^2 $$
$$ A = \pi \cdot 0.0025 $$
$$ A \approx 0.00785 \ \text{m}^2 $$
Substitute into the force equation:
$$ F = 1.01 \times 10^5 \cdot 0.00785 $$
$$ F = 793 \ \text{Pa} \ \text{m}^2 $$
Since \( 1 \ \text{N} = 1 \ \text{Pa} \cdot \text{m}^2 \):
$$ F \approx 793 \ \text{N} $$
Therefore:
$$ F \approx 7.9 \times 10^2 \ \text{N} $$
This force is approximately equivalent to the weight of an 80 kg mass.
Note. Using \( F = m g \): $$ F = 793 \ \text{N} $$ $$ m g = 793 \ \text{N} $$ $$ m = \frac{793}{g} $$ $$ m = \frac{793}{9.81} $$ Since \( 1 \ \text{N} = 1 \text{kg} \cdot \text{m/s}^2 \): $$ m = \frac{793 \ \text{kg} \ \text{m/s}^2}{9.81 \ \text{m/s}^2} $$ $$ m = 80.8 \ \text{kg} $$
Example 2
At the top of a mountain 1800 meters above sea level, atmospheric pressure is about 20% lower. What force does the air exert on the palm of a hand, modeled for simplicity as a rectangle measuring 8.0 cm by 10 cm?
At sea level, atmospheric pressure is approximately
$$ p_0 = 1.013 \times 10^5 \ \text{Pa} $$
At 1800 m, the pressure drops by 20%. Therefore, 80% of the sea-level value remains:
$$ p = 0.80 \cdot p_0 $$
$$ p = 0.80 \cdot 1.013 \times 10^5 \ \text{Pa} $$
$$ p = 0.8104 \times 10^5 \ \text{Pa} $$
$$ p = 8.104 \times 10^4 \ \text{Pa} $$
The area of the palm is
$$ A = 8.0 \ \text{cm} \times 10 \ \text{cm} = 80 \ \text{cm}^2 $$
Convert this to square meters, using $ 1 \ \text{cm}^2 = 10^{-4} \ \text{m}^2 $
$$ A = 80 \times 10^{-4} $$
$$ A = 8.0 \times 10^{-3} \ \text{m}^2 $$
The force due to pressure is given by
$$ F = p \cdot A $$
$$ F = 8.104 \times 10^4 \ \text{Pa} \times 8.0 \times 10^{-3} \ \text{m}^2 $$
$$ F = 64.832 \times 10 \ \text{Pa} \ \text{m}^2 $$
$$ F = 6.483 \times 10^2 \ \text{Pa} \ \text{m}^2 $$
Recalling that $ 1 \ \text{N} = \text{Pa} \ \text{m}^2 $
$$ F = 6.483 \times 10^2 \ \text{N} $$
Hence, the atmospheric force acting on the palm is approximately
$$ F \approx 648 \ \text{N} $$
This is roughly equivalent to the weight of a mass of
$$ m = \dfrac{F}{g} = \dfrac{648}{9.81} \approx 66.1 \ \text{kg} $$
Thus, even at an altitude of 1800 m, the air exerts a considerable force, although less than at sea level.
The Torricelli Barometer
Atmospheric pressure is equal to the pressure exerted by a mercury column 0.760 meters high at sea level under standard atmospheric conditions.
On the basis of this result, Evangelista Torricelli constructed the first mercury barometer in 1643 to measure atmospheric pressure.
A mercury barometer consists of a cylindrical glass tube, closed at one end and completely filled with a liquid of density ( $ d $ ), which is then inverted and immersed in a reservoir containing the same liquid.
The open end of the tube remains submerged below the free surface of the liquid in the reservoir.
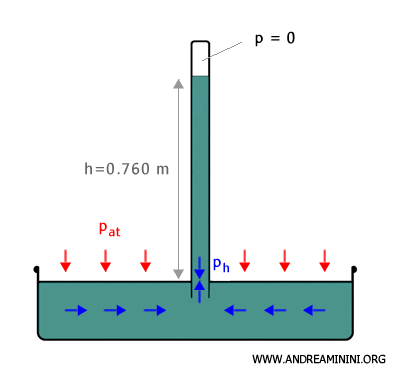
Part of the liquid flows out of the tube, leaving an empty space at the top of the column where the pressure is effectively zero. This region is known as the Torricellian vacuum.
At equilibrium, at sea level, the mercury column in the tube reaches a height ( $ h $ ) of 0.760 meters, that is 760 millimeters.
Any variation in atmospheric pressure ( $ p_{at} $ ) acts on the free surface of the liquid in the reservoir and produces a corresponding variation in the height ( $ h $ ) of the column inside the tube.
The height of the column is therefore directly proportional to the atmospheric pressure.
Note. For this reason, one of the commonly used units of pressure is the millimeter of mercury (mmHg). In particular, standard atmospheric pressure is defined as: $$ p_{at} = 1 \ atm = 760 \ mmHg $$ where $ Hg $ is the chemical symbol for mercury and $ atm $ denotes the unit of pressure called the “atmosphere”.
Derivation
According to Stevin’s law, the pressure in a fluid at equilibrium is given by
$$ p_h = p_{s} + dgh $$
where $ p_h $ is the pressure of the liquid at depth $ h $, $ p_s $ is the pressure at the upper surface of the column, $ d $ is the density of the liquid, $ g $ is the acceleration due to gravity, and $ h $ is the height of the liquid column.
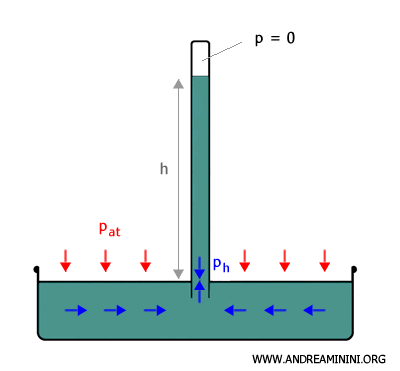
In this case, there is a vacuum at the top of the column, so the pressure at the upper surface is approximately zero $ p_{s} = 0 $
$$ p_h = 0 + dgh = dgh $$
Note. The approximation is justified by the presence of a very small mercury vapor pressure in the Torricellian vacuum, which is negligible at this level of analysis.
Consider a point in the mercury column inside the tube at the same height as the free surface of the mercury in the reservoir.
In a fluid at equilibrium, any two points at the same height are at the same pressure. Therefore, the pressure at that point $ (p_h) $ is equal to the atmospheric pressure ( $ p_{at} $ ):
$$ p_{at} = p_h $$
Since $ p_h = dgh $, it follows that
$$ p_{at} = dgh $$
The liquid in the reservoir is mercury ( Hg ), which has a known density of $ d=1.36 \cdot 10^4 \ kg/m^3 $
$$ p_{at} = (1.36 \cdot 10^4 \ kg \ m^{-3} ) \cdot gh $$
The average acceleration due to gravity at sea level is $ g=9.81 \ m/s^2 $, which can also be written as $ g=9.81 \ N/kg $
$$ p_{at} = (1.36 \cdot 10^4 \ kg \ m^{-3} ) \cdot (9.81 \ N \ kg^{-1}) h $$
Canceling the kg units gives
$$ p_{at} = (1.36 \cdot 10^4 \cdot 9.81 ) \ N \ m^{-3} h $$
Atmospheric pressure is $ p_{at} = 1.013 \cdot 10^5 \ Pa $
$$ 1.013 \cdot 10^5 \ Pa = (1.36 \cdot 10^4 \cdot 9.81 ) \ N \ m^{-3} h $$
Solving for the height $ h $ gives
$$ h = \frac{ 1.013 \cdot 10^5 \ Pa } { (1.36 \cdot 10^4 \cdot 9.81 ) \ N \ m^{-3} } $$
Simplifying the powers of 10, since $ \frac{10^5}{10^4} = 10^{5-4} = 10^1 $
$$ h = \frac{ 1.013 \cdot 10 \ Pa } { (1.36 \cdot 9.81 ) \ N \ m^{-3} } $$
Evaluating the numerical value $ \frac{1.013 \cdot 10}{1.36 \cdot 9.81} \approx 0.760 $
$$ h = 0.760 \ \frac{ Pa }{ N m^{-3} } $$
Since $ 1 \ Pa = N/m^2 $, that is $ Pa = N m^{-2} $
$$ h = 0.760 \ \frac{ N m^{-2} }{ N m^{-3} } $$
Canceling newtons (N) and simplifying the powers of meters gives $ \frac{m^{-2}}{m^{-3}} = m^{-2-(-3)} = m^{-2+3} = m^1 $
$$ h = 0.760 \ m $$
The height of the column is therefore 0.760 meters, that is 760 millimeters, as expected.
And similarly for other cases.
